Companion Animal Drug Development
Our Services
Novel Compound Veterinary Drug Development
C.S. Group has recognized that the benefits of Taiwanofungus camphoratus extend beyond human wellness, demonstrating immense potential in the field of pet healthcare. To align with PIC/S GMP international pharmaceutical manufacturing standards and explore the pharmacological applications of its bioactive compounds, the Group invested nearly NT$100 million in 2025 to establish a dedicated Antcin A Drug Research Laboratory.
Our strategic approach to pet drug development focuses on the high-demand markets associated with the top ten most common pet diseases. Specifically, we are targeting the global pet steroid market, which is valued at over US$10 billion annually. Our mission is to provide a fundamental solution that addresses the physical strain and severe side effects caused by long-term steroid use in pets, offering a safer and more effective therapeutic alternative.
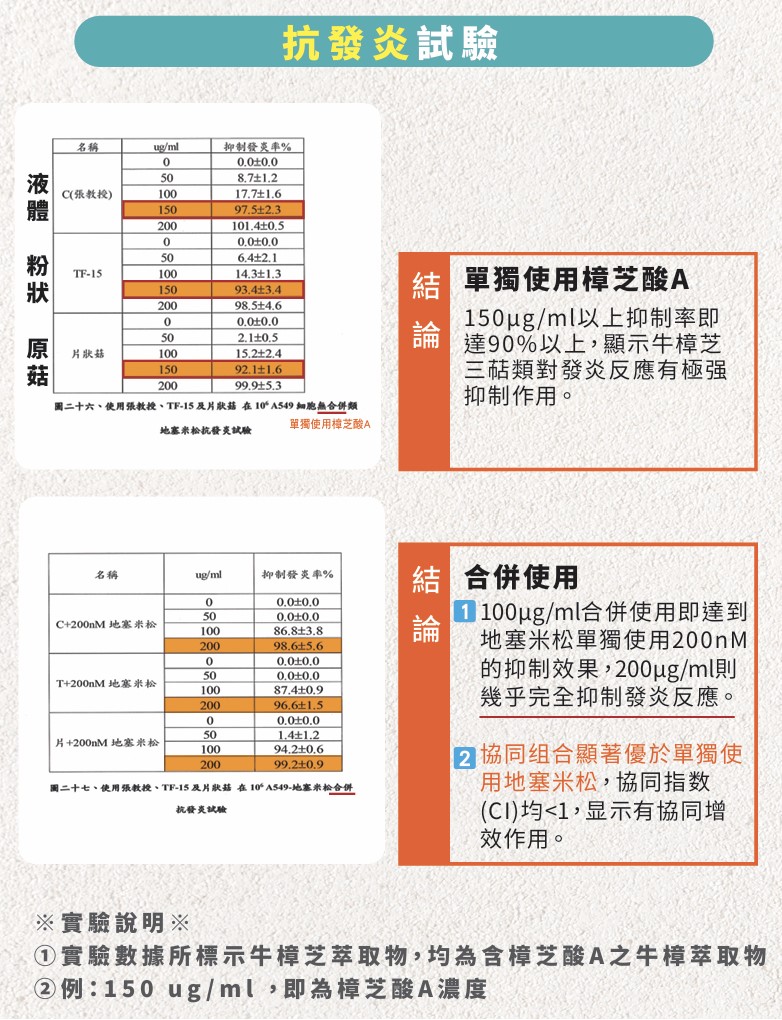
Laboratory results have confirmed that Antcin A exhibits anti-inflammatory properties comparable to those of steroids. Subsequently, our proprietary TF-15 raw material (containing high concentrations of Antcin A) was combined with the steroid Dexamethasone (DXMS) for lung cancer cell line experiments. Preliminary results demonstrate a significant synergistic effect. The experimental validations are summarized as follows:
Species Authentication: DNA sequencing of the TF-15 raw material has definitively identified it as the Taiwanofungus camphoratus species.
Grade Equivalence: Analytical testing confirms that TF-15 is equivalent in grade to high-quality Taiwanofungus camphoratus fruiting bodies cultivated via traditional wood-log methods.
Advanced Extraction Process: Utilizing a dual-extraction method—aqueous extraction to remove impurities followed by ethanol extraction—we have successfully obtained high-purity Antcin A from TF-15.
Synergistic Potential: The combination of Antcin A and Dexamethasone has been proven to possess the potential for a synergistic effect in inhibiting and reversing cancer cell progression.
The global pet anti-inflammatory drug market is projected to reach an estimated USD 4.5–5 billion by the 2030–2034 period, growing at a Compound Annual Growth Rate (CAGR) of 6.5% to 7%.
The global companion animal pain management market is projected to reach USD 2 billion by 2025 and is expected to grow to USD 3.4 billion by 2034, representing a Compound Annual Growth Rate (CAGR) of 6.2%.
The global companion animal pharmaceutical market is projected to reach USD 76.5 billion by the 2034–2037 period, expanding at a Compound Annual Growth Rate (CAGR) of 9.7%.
High-purity Antcin A Standard with a purity exceeding 95%
The Advantages of Initiating Veterinary New Drug Trials as the Primary Entry Point for Taiwanofungus camphoratus Include:
Optimized Formulations: Allows for the development of pet-specific delivery systems (e.g., oral liquids/chews) based on proven safety data.
Clinical Acceleration: Provides a critical data bridge that shortens the trajectory for human clinical trials.
Market Synergy: Scientific insights can be cross-leveraged to boost the credibility of our pet and human supplement lines.

Targeting the Multi-Billion Dollar Global Market for Pet Corticosteroids
According to data from the market research firm Valuates Reports, the global market for steroids in pet healthcare reached US$38.3 billion in 2021 and continues to show steady annual growth. Locally, the Taiwan market for veterinary steroid injectables exceeded NT$1.5 billion in 2022, with a projected Compound Annual Growth Rate (CAGR) of 5.2% over the next five years. Industry evidence confirms that Taiwanofungus camphoratus can mitigate the side effects associated with traditional steroid use. Integrating this into a combined therapy with veterinary steroids is expected to unlock significant market value.
In 2025, C.S. Group officially proposed a novel combination therapy—Antcin A in conjunction with steroids—and initiated the application for a New Compound Veterinary Drug Certificate in Taiwan. The first phase, involving background data review and pharmacokinetic (PK) testing, was successfully completed in mid-2025. We are on schedule to launch the second phase, toxicological safety testing, in the first quarter of 2026.
Industry-Academia Collaborative R&D Project for Taiwanofungus camphoratus

Pharmacological Assessment of Antcin A-Enriched Taiwanofungus camphoratus Extract in Steroidal Anti-inflammatory and Anti-tumor Activity

FAQ
Why Target the Global Pet Steroid Market with Taiwanofungus camphoratus Extract?
Traditional new drug development for human use is notoriously time-consuming and capital-intensive, requiring massive investment in animal and human clinical trials with no guarantee of success. In contrast, Taiwanofungus camphoratus benefits from a rich and comprehensive foundation of existing research within the industry, eliminating the need to start from the ground up.
International medical research has consistently shown that long-term use of synthetic steroids in both humans and animals leads to significant drug dependency and a host of side effects, notably imposing a heavy burden on liver and kidney functions. Conversely, AC is recognized for its hepatoprotective and nephroprotective (liver and kidney-protecting) properties. Further research into its specific bioactive compounds—Antcins A, B, C, H, and K—has revealed that Antcin A is the only compound capable of successfully translocating into the cell nucleus to trigger biological action. It functions as a natural glucocorticoid mimetic, delivering anti-inflammatory effects equivalent to those of steroids.
By leveraging these findings, we are developing a synergistic combination therapy that pairs AC with traditional corticosteroids. This fixed-dose combination (FDC) will be prioritized for the veterinary pharmaceutical market, accelerating its practical application and creating immediate clinical and commercial value.
Beyond anti-inflammatories, what other veterinary pharmaceutical markets can Antcin A be applied to?
The current global veterinary pharmaceutical landscape is segmented into several key therapeutic areas, including ectoparasiticides (external deworming), endoparasiticides (internal deworming), endectocides (all-in-one parasiticides), atopic dermatitis, anti-inflammatory and analgesics, antiemetics, insulins, antibiotics, and congestive heart failure (CHF) medications.
Antcin A, a naturally occurring steroidal compound, possesses potent anti-inflammatory properties without the severe side effects typically associated with conventional synthetic steroids. Given its superior safety profile and efficacy, Antcin A is poised to capture significant market share and create extensive applications across various sectors of the veterinary pharmaceutical industry.
What are the future market deployment plans for fixed-dose combination (FDC) therapies involving Antcin A and corticosteroids?
We will actively engage leading research institutions across Asia and the United States, along with pharmaceutical partners and veterinary healthcare organizations, to explore collaborative studies and long-term partnerships. Through these efforts, we aim to introduce Taiwan’s premium, naturally derived anti-inflammatory bioactive compounds to more veterinarians and pet owners worldwide.

